Abstract
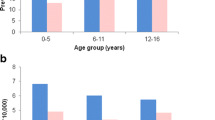
Nationwide analyses of drug use can provide a prevalence estimate of the underlying disease and can help in understanding the characteristics of treatment. This study aimed for such analyses regarding the utilization of antiepileptic drugs (AED) for epilepsy in Germany. In 2009, all 4,115,705 AED prescriptions of all German patients with statutory health insurance (70,011,508 persons) were retrospectively analyzed. The IMS® LRx database served as data source, which accesses nationwide pharmacy data centers processing all German prescription data. To establish the age and sex-specific percentage of patients taking AED because of epilepsy, we used a second database, Disease Analyzer®, which covered a representative sample of the German population (7.2 million patients) and contained ICD10 codes alongside with prescription data. The period prevalence of patients taking AED because of epilepsy was 9.1/1,000 (children/adolescents: 5.2/1,000; elderly: 12.5/1,000). Of the patients, 83.1 % took at least one of four AED: valproate (29.8 %), carbamazepine (26.4 %), lamotrigine (21.4 %), and levetiracetam (16.9 %). Oxcarbazepine and sultiame were popular with pediatricians. Elderly patients frequently received phenytoin and primidone. More than half of the patients were treated by family physicians; 68 % took AED in monotherapy and 7.9 % received >2 AED (children/adolescents: 12.5 %). The costs for AED prescribed for epilepsy amounted to €285.1 Mio (median AED costs/patient: €158/a). The German 2009 prevalence of epileptic patients taking AED was 9.1/1,000. Family physicians cared for the majority of patients. Prevalence and prescribing patterns changed with age. Costs of AED against epilepsy added up to 1 % of total medication costs in Germany.
Similar content being viewed by others
References
Alacqua M, Trifiro G, Spina E, Moretti S, Tari DU, Bramanti P, Caputi AP, Arcoraci V (2009) Newer and older antiepileptic drug use in southern Italy: a population-based study during the years 2003–2005. Epilepsy Res 85:107–113
Andermann F, Duh MS, Gosselin A, Paradis PE (2007) Compulsory generic switching of antiepileptic drugs: high switchback rates to branded compounds compared with other drug classes. Epilepsia 48:464–469
Banerjee PN, Filippi D, Allen HW (2009) The descriptive epidemiology of epilepsy–a review. Epilepsy Res 85:31–45
Banfi R, Borselli G, Marinai C, Borgheresi A, Cavalieri A (1995) Epidemiological study of epilepsy by monitoring prescriptions of antiepileptic drugs. Pharm World Sci 17:138–140
Becher H, Kostev K, Schroder-Bernhardi D (2009) Validity and representativeness of the “Disease Analyzer” patient database for use in pharmacoepidemiological and pharmacoeconomic studies. Int J Clin Pharmacol Ther 47:617–626
Beghi E, Garattini L, Ricci E, Cornago D, Parazzini F (2004) Direct cost of medical management of epilepsy among adults in Italy: a prospective cost-of-illness study (EPICOS). Epilepsia 45:171–178
Beghi M, Savica R, Beghi E, Nobili A, Garattini L (2009) Utilization and costs of antiepileptic drugs in the elderly: still an unsolved issue. Drugs Aging 26:157–168
Bielen I, Sruk A, Planjar-Prvan M, Cvitanovic-Sojat L, Kosicek M, Bergman-Markovic B, Baraba R, Butkovic-Soldo S (2009) Age-related pattern of the antiepileptic drug utilization in active epilepsy: a population-based survey. Coll Antropol 33:659–663
Duh MS, Paradis PE, Latremouille-Viau D, Greenberg PE, Lee SP, Durkin MB, Wan GJ, Rupnow MF, LeLorier J (2009) The risks and costs of multiple-generic substitution of topiramate. Neurology 72:2122–2129
Ekman M, Forsgren L (2004) Economic evidence in epilepsy: a review. Eur J Health Econ 5(Suppl 1):S36–S42
Faught E, Richman J, Martin R, Funkhouser E, Foushee R, Kratt P, Kim Y, Clements K, Cohen N, Adoboe D, Knowlton R, Pisu M (2012) Incidence and prevalence of epilepsy among older US Medicare beneficiaries. Neurology 78:448–453
Forsgren I, Beghi E, Ekman M (2005) Cost of epilepsy in Europe. Eur J Neurol 12(Suppl 1):54–58
Forsgren L, Beghi E, Oun A, Sillanpaa M (2005) The epidemiology of epilepsy in Europe—a systematic review. Eur J Neurol 12:245–253
Gissler M, Jarvelin MR, Louhiala P, Hemminki E (1999) Boys have more health problems in childhood than girls: follow-up of the 1987 Finnish birth cohort. Acta Paediatr 88:310–314
Gustavsson A, Svensson M, Jacobi F, Allgulander C, Alonso J, Beghi E, Dodel R, Ekman M, Faravelli C, Fratiglioni L, Gannon B, Jones DH, Jennum P, Jordanova A, Jonsson L, Karampampa K, Knapp M, Kobelt G, Kurth T, Lieb R, Linde M, Ljungcrantz C, Maercker A, Melin B, Moscarelli M, Musayev A, Norwood F, Preisig M, Pugliatti M, Rehm J, Salvador-Carulla L, Schlehofer B, Simon R, Steinhausen HC, Stovner LJ, Vallat JM, den Bergh PV, van OJ, Vos P, Xu W, Wittchen HU, Jonsson B, Olesen J (2011) Cost of disorders of the brain in Europe 2010. Eur Neuropsychopharmacol 21:718–779
Hamer HM, Spottke A, Aletsee C, Knake S, Reis J, Strzelczyk A, Oertel WH, Rosenow F, Dodel R (2006) Direct and indirect costs of refractory epilepsy in a tertiary epilepsy center in Germany. Epilepsia 47:2165–2172
Heaney DC, Sander JW (2007) Antiepileptic drugs: generic versus branded treatments. Lancet Neurol 6:465–468
Helmers SL, Paradis PE, Manjunath R, Duh MS, Lafeuille MH, Latremouille-Viau D, Lefebvre P, Labiner DM (2010) Economic burden associated with the use of generic antiepileptic drugs in the United States. Epilepsy Behav 18:437–444
Holden EW, Grossman E, Nguyen HT, Gunter MJ, Grebosky B, Von WA, Nelson L, Robinson S, Thurman DJ (2005) Developing a computer algorithm to identify epilepsy cases in managed care organizations. Dis Manag 8:1–14
Hollingworth SA, Eadie MJ (2010) Antiepileptic drugs in Australia: 2002–2007. Pharmacoepidemiol Drug Saf 19:82–89
Hsieh LP, Huang CY (2009) Antiepileptic drug utilization in Taiwan: analysis of prescription using National Health Insurance database. Epilepsy Res 84:21–27
Johannessen LC, Larsson PG, Rytter E, Johannessen SI (2009) Antiepileptic drugs in epilepsy and other disorders–a population-based study of prescriptions. Epilepsy Res 87:31–39
Kesselheim AS, Stedman MR, Bubrick EJ, Gagne JJ, Misono AS, Lee JL, Brookhart MA, Avorn J, Shrank WH (2010) Seizure outcomes following the use of generic versus brand-name antiepileptic drugs: a systematic review and meta-analysis. Drugs 70:605–621
Kramer G, Biraben A, Carreno M, Guekht A, de Haan GJ, Jedrzejczak J, Josephs D, RK van, Zaccara G (2007) Current approaches to the use of generic antiepileptic drugs. Epilepsy Behav 11:46–52
Labiner DM, Paradis PE, Manjunath R, Duh MS, Lafeuille MH, Latremouille-Viau D, Lefebvre P, Helmers SL (2010) Generic antiepileptic drugs and associated medical resource utilization in the United States. Neurology 74:1566–1574
Mantel-Teeuwisse AK, Klungel OH, Verschuren WM, Porsius A, BA de (2001) Comparison of different methods to estimate prevalence of drug use by using pharmacy records. J Clin Epidemiol 54:1181–1186
Marson AG, Al Kharusi AM, Alwaidh M, Appleton R, Baker GA, Chadwick DW, Cramp C, Cockerell OC, Cooper PN, Doughty J, Eaton B, Gamble C, Goulding PJ, Howell SJ, Hughes A, Jackson M, Jacoby A, Kellett M, Lawson GR, Leach JP, Nicolaides P, Roberts R, Shackley P, Shen J, Smith DF, Smith PE, Smith CT, Vanoli A, Williamson PR (2007) The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomised controlled trial. Lancet 369:1000–1015
Marson AG, Al Kharusi AM, Alwaidh M, Appleton R, Baker GA, Chadwick DW, Cramp C, Cockerell OC, Cooper PN, Doughty J, Eaton B, Gamble C, Goulding PJ, Howell SJ, Hughes A, Jackson M, Jacoby A, Kellett M, Lawson GR, Leach JP, Nicolaides P, Roberts R, Shackley P, Shen J, Smith DF, Smith PE, Smith CT, Vanoli A, Williamson PR (2007) The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalised and unclassifiable epilepsy: an unblinded randomised controlled trial. Lancet 369:1016–1026
Oles KS, Gal P, Penry JK, Tapscott WK (1987) Use of antiepileptic drugs in the elderly population. Public Health Rep 102:335–337
Oun A, Haldre S, Magi M (2006) Use of antiepileptic drugs in Estonia: an epidemiologic study of adult epilepsy. Eur J Neurol 13:465–470
Pugliatti M, Beghi E, Forsgren L, Ekman M, Sobocki P (2007) Estimating the cost of epilepsy in Europe: a review with economic modeling. Epilepsia 48:2224–2233
Roberts SJ, Feely M, Bateman DN (1998) Prescribing of anti-epileptic drugs in the northern and Yorkshire region: 1992–1995. Seizure 7:127–132
Rochat P, Hallas J, Gaist D, Friis ML (2001) Antiepileptic drug utilization: a Danish prescription database analysis. Acta Neurol Scand 104:6–11
Sancho J, Pena P, Rufo M, Palacios G, Masramon X, Rejas J (2008) Health and non-health care resources use in the management of adult outpatients with drug-resistant epilepsy in Spain: a cost-of-illness study (LINCE study). Epilepsy Res 81:176–187
Savica R, Beghi E, Mazzaglia G, Innocenti F, Brignoli O, Cricelli C, Caputi AP, Musolino R, Spina E, Trifiro G (2007) Prescribing patterns of antiepileptic drugs in Italy: a nationwide population-based study in the years 2000–2005. Eur J Neurol 14:1317–1321
Schachter SC, Cramer GW, Thompson GD, Chaponis RJ, Mendelson MA, Lawhorne L (1998) An evaluation of antiepileptic drug therapy in nursing facilities. J Am Geriatr Soc 46:1137–1141
Shackleton DP, Westendorp RG, Kasteleijn-Nolst Trenite DG, de BA, Herings RM (1997) Dispensing epilepsy medication: a method of determining the frequency of symptomatic individuals with seizures. J Clin Epidemiol 50:1061–1068
Strzelczyk A, Reese JP, Dodel R, Hamer HM (2008) Cost of epilepsy: a systematic review. Pharmacoeconomics 26:463–476
Tetto A, Manzoni P, Millul A, Beghi E, Garattini L, Tartara A, Avanzini G (2002) The costs of epilepsy in Italy: a prospective cost-of-illness study in referral patients with disease of different severity. Epilepsy Res 48:207–216
Tsiropoulos I, Gichangi A, Andersen M, Bjerrum L, Gaist D, Hallas J (2006) Trends in utilization of antiepileptic drugs in Denmark. Acta Neurol Scand 113:405–411
van de Vrie-Hoekstra NW, de Vries TW, van den Berg PB, Brouwer OF, de Jong-van den Berg LT (2008) Antiepileptic drug utilization in children from 1997 to 2005–a study from the Netherlands. Eur J Clin Pharmacol 64:1013–1020
Waaler PE, Blom BH, Skeidsvoll H, Mykletun A (2000) Prevalence, classification, and severity of epilepsy in children in western Norway. Epilepsia 41:802–810
Conflicts of interest
The authors report the following possible sources of conflicts of interest. Dr. Hamer has served on the scientific advisory board of Eisai, Pfizer, and UCB Pharma. He served on the speakers’ bureau of Desitin, Eisai, GlaxoSmithKline, and UCB Pharma and received research funding from Desitin, Janssen-Cilag GlaxoSmithKline, and UCB Pharma. Dr. Strzelczyk has served on the speakers’ bureau of Eisai and received funding for travel to present findings from Eisai, Pfizer and UCB Pharma. K. Kostev is an employee of IMS HEALTH, a company that focuses on analyses of pharmacy records and runs the database used in this analysis. This study, however, was not part of any business project. Dr. Knake has served on the scientific advisory board of UCB Pharma. She served on the speakers’ bureau of Eisai, GlaxoSmithKline, and UCB Pharma. Dr. Rosenow has served on the scientific advisory board of Eisai, GlaxoSmithKline, Pfizer, and UCB Pharma. He served on the speakers’ bureau of Eisai, GlaxoSmithKline, and UCB Pharma and received research funding from Novartis and UCB Pharma. Dr. Dodel has served on the scientific advisory board of Lilly. He served on the speakers’ bureau of Baxter, Boehringer Ingelheim, CSL Behring, Eisai, GlaxoSmithKline, Lundbeck, Merz, Octapharma, Orion Pharma, Pfizer, Solvay, and UCB Pharma and received research funding from CSL Behring, Lundbeck, Novartis, Rentschler, and ZLB Behring. He received honoraria for consultancy from Affiris, Baxter, GE Healthcare, and Octapharma. The remaining authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamer, H.M., Dodel, R., Strzelczyk, A. et al. Prevalence, utilization, and costs of antiepileptic drugs for epilepsy in Germany—a nationwide population-based study in children and adults. J Neurol 259, 2376–2384 (2012). https://doi.org/10.1007/s00415-012-6509-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6509-3